Chinese Journal of Tissue Engineering Research ›› 2014, Vol. 18 ›› Issue (51): 8269-8274.doi: 10.3969/j.issn.2095-4344.2014.51.013
Previous Articles Next Articles
Effect of Kruppel-like factor 6 overexpression on cholesterol accumulation in macrophages
Wang Hui-qing, Zhang Zong-qi, Huang Wen-hong, Wang Xiao-fei, Zhu Yan
- Department of Electrocardiogram, No. 3 People’s Hospital Affiliated to Shanghai Jiao Tong University School of Medicine, Shanghai 201999, China
-
Online:2014-12-10Published:2014-12-10 -
Contact:Zhu Yan, Attending physician, Department of Electrocardiogram, No. 3 People’s Hospital Affiliated to Shanghai Jiao Tong University School of Medicine, Shanghai 201999, China -
About author:Wang Hui-qing, Physician, Department of Electrocardiogram, No. 3 People’s Hospital Affiliated to Shanghai Jiao Tong University School of Medicine, Shanghai 201999, China -
Supported by:the National Science Fund for Distinguished Young Scholars, No. 81200206; the Project of Shanghai Health Bureau, No. 2010073
CLC Number:
Cite this article
Wang Hui-qing, Zhang Zong-qi, Huang Wen-hong, Wang Xiao-fei, Zhu Yan. Effect of Kruppel-like factor 6 overexpression on cholesterol accumulation in macrophages[J]. Chinese Journal of Tissue Engineering Research, 2014, 18(51): 8269-8274.
share this article
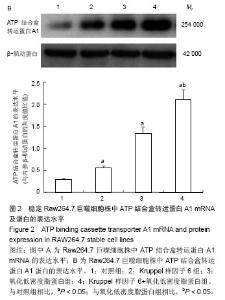
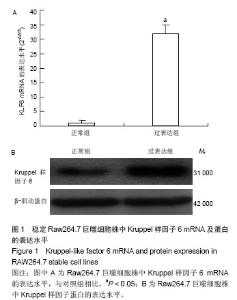
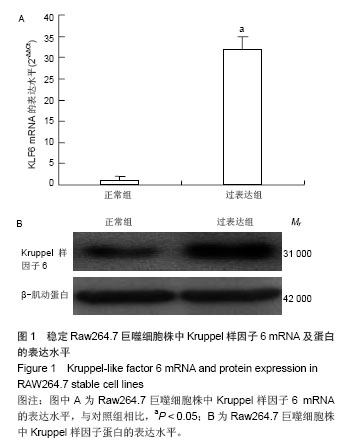
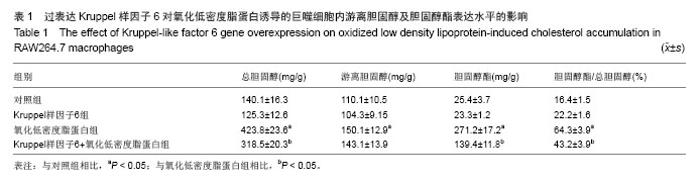
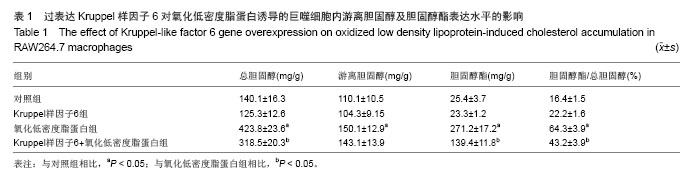
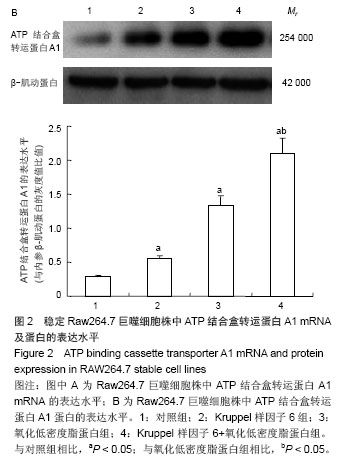
2.3 过表达Kruppel样因子6对巨噬细胞ATP结合盒转运蛋白A1 mRNA和蛋白表达的影响 实时PCR结果显示,与对照组相比,Kruppel样因子6组巨噬细胞ATP结合盒转运蛋白A1 mRNA表达增加(P < 0.05);而与对照组相比,氧化低密度脂蛋白组巨噬细胞ATP结合盒转运蛋白A1 mRNA的表达水平明显上升(P < 0.05),且Kruppel样因子6+氧化低密度脂蛋白组细胞ATP结合盒转运蛋白A1 mRNA的表达水平较氧化低密度脂蛋白组显著增加 (P < 0.05;图2A)。并且Western blot结果显示,各组巨噬细胞ATP结合盒转运蛋白A1蛋白表达改变与mRNA上调变化一致。Kruppel样因子6+氧化低密度脂蛋白组巨噬细胞ATP结合盒转运蛋白A1蛋白表达较氧化低密度脂蛋白组细胞显著升高(P < 0.05;图2B)。"
| [1]Moore KJ, Tabas I. Macrophages in the pathogenesis of atherosclerosis. Cell. 2011;145(3): 341-355. [2]Bobryshev YV. Monocyte recruitment and foam cell formation in atherosclerosis. Micron. 2006;37(3):208-222. [3]Taghavie-Moghadam PL, Butcher MJ, Galkina EV. The dynamic lives of macrophage and dendritic cell subsets in atherosclerosis. Ann N Y Acad Sci. 2014;1319:19-37. [4]Botham KM, Wheeler-Jones CP. Postprandial lipoproteins and the molecular regulation of vascular homeostasis. Prog Lipid Res. 2013;52(4):446-464. [5]Imanishi T, Akasaka T. Novel strategies to target inflammatory processes in atherosclerosis. Curr Pharm Des. 2013;19(9): 1616-1625. [6]Webb NR, Moore KJ. Macrophage-derived foam cells in atherosclerosis: lessons from murine models and implications for therapy. Curr Drug Targets. 2007;8(12):1249-1263. [7]Fernández-Velasco M, González-Ramos S, Boscá L. Involvement of monocytes/macrophages as key factors in the development and progression of cardiovascular diseases. Biochem J. 2014;458(2):187-193. [8]Gui T, Shimokado A, Sun Y, et al. Diverse roles of macrophages in atherosclerosis: from inflammatory biology to biomarker discovery. Mediators Inflamm. 2012;2012:693083. [9]Yuan Y, Li P, Ye J. Lipid homeostasis and the formation of macrophage-derived foam cells in atherosclerosis. Protein Cell. 2012;3(3):173-181. [10]Andreoli V, Gehrau RC, Bocco JL. Biology of Krüppel-like factor 6 transcriptional regulator in cell life and death. IUBMB Life. 2010;62(12):896-905. [11]Zhang Y, Lei CQ, Hu YH, et al. Krüppel-like factor 6 is a co-activator of NF-κB that mediates p65-dependent transcription of selected downstream genes. J Biol Chem. 2014;289(18):12876-12885. [12]Qi W, Holian J, Tan CY, et al. The roles of Kruppel-like factor 6 and peroxisome proliferator-activated receptor-γ in the regulation of macrophage inflammatory protein-3α at early onset of diabetes. Int J Biochem Cell Biol. 2011;43(3): 383-392. [13]Duan SZ, Usher MG, Mortensen RM. Peroxisome proliferator-activated receptor-gamma-mediated effects in the vasculature. Circ Res. 2008;102(3):283-294. [14]Mao Z, Ong AC. Peroxisome proliferator-activated receptor gamma agonists in kidney disease--future promise, present fears. Nephron Clin Pract. 2009;112(4):c230-241. [15]Ketsawatsomkron P, Pelham CJ, Groh S, et al. Does peroxisome proliferator-activated receptor-gamma (PPAR gamma) protect from hypertension directly through effects in the vasculature? J Biol Chem. 2010;285(13):9311-9316. [16]Chen YC, Wu JS, Tsai HD, et al. Peroxisome proliferator-activated receptor gamma (PPAR-γ) and neurodegenerative disorders. Mol Neurobiol. 2012;46(1): 114-124. [17]Freitag CM, Miller RJ. Peroxisome proliferator-activated receptor agonists modulate neuropathic pain: a link to chemokines? Front Cell Neurosci. 2014;8:238. [18]Yang J, Zhou Y, Guan Y. PPARγ as a therapeutic target in diabetic nephropathy and other renal diseases. Curr Opin Nephrol Hypertens. 2012;21(1):97-105. [19]Date D, Das R, Narla G, et al. Kruppel-like transcription factor 6 regulates inflammatory macrophage polarization. J Biol Chem. 2014;289(15):10318-10329. [20]Steinberg D. Modified forms of low-density lipoprotein and atherosclerosis. J Intern Med. 1993;233(3):227-232. [21]Lei L, Xiong Y, Chen J, et al. TNF-alpha stimulates the ACAT1 expression in differentiating monocytes to promote the CE-laden cell formation. J Lipid Res. 2009;50(6): 1057-1067. [22]Luo ZF, Feng B, Mu J, et al. Effects of 4-phenylbutyric acid on the process and development of diabetic nephropathy induced in rats by streptozotocin: regulation of endoplasmic reticulum stress-oxidative activation. Toxicol Appl Pharmacol. 2010;246(1-2):49-57. [23]Glass CK, Witztum JL. Atherosclerosis. the road ahead. Cell. 2001;104(4):503-516. [24]Hansson GK. Inflammation, atherosclerosis, and coronary artery disease. N Engl J Med. 2005;352(16):1685-1695. [25]Mehta JL, Saldeen TG, Rand K. Interactive role of infection, inflammation and traditional risk factors in atherosclerosis and coronary artery disease. J Am Coll Cardiol. 1998;31(6): 1217-1225. [26]Steinberg D. Hypercholesterolemia and inflammation in atherogenesis: two sides of the same coin. Mol Nutr Food Res. 2005;49(11):995-998. [27]Malkin CJ, Pugh PJ, Jones RD, et al. Testosterone as a protective factor against atherosclerosis--immunomodulation and influence upon plaque development and stability. J Endocrinol. 2003;178(3):373-380. [28]George JF, Pinderski LJ, Litovsky S, et al. Of mice and men: mouse models and the molecular mechanisms of post-transplant coronary artery disease. J Heart Lung Transplant. 2005;24(12):2003-2014. [29]Li JJ. Inflammation: an important mechanism for different clinical entities of coronary artery diseases. Chin Med J (Engl). 2005;118(21):1817-1826. [30]O'Kelly BF, Massie BM, Tubau JF, et al. Coronary morbidity and mortality, pre-existing silent coronary artery disease, and mild hypertension. Ann Intern Med. 1989;110(12):1017-1026. [31]Bellosta S, Bernini F, Chinetti G, et al. Macrophage function and stability of the atherosclerotic plaque: progress report of a European project. Nutr Metab Cardiovasc Dis. 2002;12(1): 3-11. [32]Granada JF, Kaluza GL, Wilensky RL, et al. Porcine models of coronary atherosclerosis and vulnerable plaque for imaging and interventional research. EuroIntervention. 2009;5(1): 140-148. [33]Li AC, Glass CK. The macrophage foam cell as a target for therapeutic intervention. Nat Med. 2002;8(11):1235-1242. [34]Hansson GK, Libby P. The immune response in atherosclerosis: a double-edged sword. Nat Rev Immunol. 2006;6(7):508-519. [35]Hansson GK, Nilsson J. Vaccination against atherosclerosis? Induction of atheroprotective immunity. Semin Immunopathol. 2009;31(1):95-101. [36]Nilsson J, Nordin Fredrikson G, Schiopu A, et al. Oxidized LDL antibodies in treatment and risk assessment of atherosclerosis and associated cardiovascular disease. Curr Pharm Des. 2007;13(10):1021-1030. [37]Loppnow H, Werdan K, Buerke M. Vascular cells contribute to atherosclerosis by cytokine- and innate-immunity-related inflammatory mechanisms. Innate Immun. 2008;14(2):63-87. [38]Meier P, Meier R, Blanc E. Influence of CD4+/CD25+ regulatory T cells on atherogenesis in patients with end-stage kidney disease. Expert Rev Cardiovasc Ther. 2008;6(7): 987-997. [39]Gibson FC 3rd, Ukai T, Genco CA. Engagement of specific innate immune signaling pathways during Porphyromonas gingivalis induced chronic inflammation and atherosclerosis. Front Biosci. 2008;13:2041-2059. [40]Nilsson J, Fredrikson GN, Björkbacka H, et al. Vaccines modulating lipoprotein autoimmunity as a possible future therapy for cardiovascular disease. J Intern Med. 2009;266(3): 221-231. [41]Bechmann LP, Vetter D, Ishida J, et al. Post-transcriptional activation of PPAR alpha by KLF6 in hepatic steatosis. J Hepatol. 2013;58(5):1000-1006. [42]Narla G, Friedman SL, Martignetti JA. Krüppel cripples prostate cancer: KLF6 progress and prospects. Am J Pathol. 2003;162(4):1047-1052. [43]Weber U, Rodriguez E, Martignetti J, et al. Luna, a Drosophila KLF6/KLF7, is maternally required for synchronized nuclear and centrosome cycles in the preblastoderm embryo. PLoS One. 2014;9(6):e96933. [44]Ozdemir F, Koksal M, Ozmen V, et al. Mutations and Krüppel-like factor 6 (KLF6) expression levels in breast cancer. Tumour Biol. 2014;35(6):5219-5225. [45]Das H, Kumar A, Lin Z, et al. Kruppel-like factor 2 (KLF2) regulates proinflammatory activation of monocytes. Proc Natl Acad Sci U S A. 2006;103(17):6653-6658. [46]Nayak L, Goduni L, Takami Y, et al. Kruppel-like factor 2 is a transcriptional regulator of chronic and acute inflammation. Am J Pathol. 2013;182(5):1696-1704. [47]Feinberg MW, Cao Z, Wara AK, et al. Kruppel-like factor 4 is a mediator of proinflammatory signaling in macrophages. J Biol Chem. 2005;280(46):38247-38258. [48]Liao X, Sharma N, Kapadia F, et al. Krüppel-like factor 4 regulates macrophage polarization. J Clin Invest. 2011;121(7): 2736-2749. [49]Joyce CW, Amar MJ, Lambert G, et al. The ATP binding cassette transporter A1 (ABCA1) modulates the development of aortic atherosclerosis in C57BL/6 and apoE-knockout mice. Proc Natl Acad Sci U S A. 2002;99(1):407-412. [50]Westerterp M, Murphy AJ, Wang M, et al. Deficiency of ATP-binding cassette transporters A1 and G1 in macrophages increases inflammation and accelerates atherosclerosis in mice. Circ Res. 2013;112(11):1456-1465. [51]Chen X, Zhao Y, Guo Z, et al. Transcriptional regulation of ATP-binding cassette transporter A1 expression by a novel signaling pathway. J Biol Chem. 2011;286(11):8917-8923. [52]Chawla A, Boisvert WA, Lee CH, et al. A PPAR gamma-LXR-ABCA1 pathway in macrophages is involved in cholesterol efflux and atherogenesis. Mol Cell. 2001;7(1): 161-171. |
| [1] | Mao Xin, Yu Limei, Wang Feng. Important role of mesenchymal stem cells in immune tolerance induction in heart transplantation [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(13): 2070-2078. |
| [2] | Li Junqi, Tian Guangzhao, Chen Mingxue, Wang Hao, Liu Shuyun, Sui Xiang, Huang Jingxiang, Li Ming, Guo Quanyi. Regulatory effect of acellular cartilage extracellular matrix on phenotype of mouse macrophage line [J]. Chinese Journal of Tissue Engineering Research, 2021, 25(10): 1512-1516. |
| [3] | Cao Jiangang, Chen Desheng. Synovial macrophages in osteoarthritis: roles and features [J]. Chinese Journal of Tissue Engineering Research, 2020, 24(29): 4731-4736. |
| [4] | Liang Min1, Wang Hainiu2, Huang Peng2, Zhu Weihua2, Li Shunchang1 . Systematic review and meta-analysis of effect of resistance exercise on glucose and lipid metabolism disorder in type 2 diabetes mellitus patients [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(35): 5718-5726. |
| [5] | Shen Qian, Hu Peixin, Zhong Shuxian, Yang Yalan, Zhang Saixia, Li Chun. Tortoise shell ointment for acute skin wound in rats via Wnt/beta-catenin signal pathway in hair follicle stem cells Tortoise shell ointment for acute skin wound in rats via Wnt/beta-catenin signal pathway in hair follicle stem cells [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(29): 4668-4674. |
| [6] | Liu Wenhua, Liang Jinfeng, Deng Shaojie. Resveratrol reduces tumor nuclear factor-α expression in wear-particle-treated macrophages by regulating the level of intracellular oxidative stress [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(26): 4115-4120. |
| [7] | Chen Bingquan, Peng Yi, Xiao Yi, Peng Zhiyong, Zhao Jiling, Yu Guolong. Effect of human umbilical cord blood mesenchymal stem cells on the transformation of M2 macrophages from mouse bone marrow [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(25): 3987-3992. |
| [8] | Liu Baoyi1, Li Minde1, Yang Fan2, Li Shaopeng1, Chen Haojie1, Xiao Peng1. Angiotensin II promotes steroid-induced avascular necrosis of the femoral head in mice [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(19): 3044-3049. |
| [9] | Song Yuxian, Zhang Dongya, Xu Yujun, Hou Yayi, Ni Yanhong. Regulatory effect of human umbilical cord mesenchymal stem cells-derived exosomes on macrophage polarization [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(13): 2002-2008. |
| [10] | Yu Hong, Liu Yan, Li Bo. Effects of high-intensity circuit training on body composition, cardiopulmonary fitness and metabolic syndrome markers in an overweight population [J]. Chinese Journal of Tissue Engineering Research, 2019, 23(11): 1738-1742. |
| [11] | Gong Dong, Zhen Ping, Wang Rong. Role of macrophage polarization in periprosthetic osteolysis: its research status and advance [J]. Chinese Journal of Tissue Engineering Research, 2018, 22(7): 1120-1125. |
| [12] | Gao Wei, Lin Zhenxun, Zhen Puxiang, Chen Yan, Kuang Xiaocong, Hua Qikai. Macrophages promote the healing of severe diabetic foot wounds after tibial transverse transport [J]. Chinese Journal of Tissue Engineering Research, 2018, 22(36): 5811-5815. |
| [13] | Zeng Ronghua, Wu Huiting, Zhou Lu, Ouyang Hougan, Gao Shuliang, Wang Jianmin, Ouyang Yanchu, Cui Tiantian. Changes of immune function of mesenteric lymph nodes in rats with spleen deficiency syndrome after acupuncture at Zusanli [J]. Chinese Journal of Tissue Engineering Research, 2018, 22(36): 5846-5851. |
| [14] | Yu Xi, Hu Jun-tao, Lai Jie, Wang Rui-zhi, Ma Lan-lan, Tang Zhan-hong. Comparison of the biological characteristics of primary rat macrophages extracted by three methods [J]. Chinese Journal of Tissue Engineering Research, 2018, 22(24): 3863-3868. |
| [15] |
Li Chong, Yan Cheng, Jin Hui-min, Wu Shui-yun, Qiang Ye-tao, Yan Nan-nan, Xiao Teng-fei, Xia Sheng.
Biocompatibility of macrophages with quantum dots
[J]. Chinese Journal of Tissue Engineering Research, 2017, 21(26): 4217-4221.
|
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||